|
The "rocking" cells used have been improved over the years. Mercury cell electrolysis, also known as the Castner-Kellner process, was the first method used at the end of the nineteenth century to produce chlorine on an industrial scale. Overall process: 2 NaCl (or KCl) + 2 H 2O → Cl 2 + H 2 + 2 NaOH (or KOH) There are three industrial methods for the extraction of chlorine by electrolysis of chloride solutions, all proceeding according to the following equations:Ĭathode: 2 H + (aq) + 2 e − → H 2 (g) Anode: 2 Cl − (aq) → Cl 2 (g) + 2 e − Chlorine can also be produced by the electrolysis of a solution of potassium chloride, in which case the co-products are hydrogen and caustic potash ( potassium hydroxide). These two products, as well as chlorine itself, are highly reactive. The production of chlorine results in the co-products caustic soda ( sodium hydroxide, NaOH) and hydrogen gas (H 2). Along with chlorine, this chloralkali process yields hydrogen gas and sodium hydroxide, according to the following chemical equation:Ģ NaCl + 2 H 2O → Cl 2 + H 2 + 2 NaOH Production Gas extractionĬhlorine can be manufactured by electrolysis of a sodium chloride solution ( brine). Industrially, elemental chlorine is usually produced by the electrolysis of sodium chloride dissolved in water. Over 2000 naturally-occurring organic chlorine compounds are known. Common chloride minerals include halite ( sodium chloride), sylvite ( potassium chloride), and carnallite (potassium magnesium chloride hexahydrate). Most chloride salts are soluble in water, thus, chloride-containing minerals are usually only found in abundance in dry climates or deep underground. Even higher concentrations of chloride are found in the Dead Sea and in underground brine deposits. In nature, chlorine is found primarily as the chloride ion, a component of the salt that is deposited in the earth or dissolved in the oceans - about 1.9% of the mass of seawater is chloride ions. 36Cl has seen use in other areas of the geological sciences, including dating ice and sediments. Thus, as an event marker of 1950s water in soil and ground water, 36Cl is also useful for dating waters less than 50 years before the present. The residence time of 36Cl in the atmosphere is about 1 week. Additionally, large amounts of 36Cl were produced by irradiation of seawater during atmospheric detonations of nuclear weapons between 19. The half-life of this hydrophilic nonreactive isotope makes it suitable for geologic dating in the range of 60,000 to 1 million years. 36Cl decays to 36 S and to 36 Ar, with a combined half-life of 308,000 years. In the subsurface environment, 36Cl is generated primarily as a result of neutron capture by 35Cl or muon capture by 40 Ca. 36Cl is produced in the atmosphere by spallation of 36 Ar by interactions with cosmic ray protons. 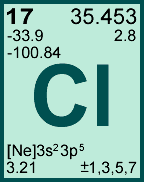
Trace amounts of radioactive 36Cl exist in the environment, in a ratio of about 7x10 −13 to 1 with stable isotopes. There are two principal stable isotopes, 35Cl (75.77%) and 37Cl (24.23%), giving chlorine atoms in bulk an apparent atomic weight of 35.4527 g/mol. In the upper atmosphere, chlorine-containing molecules have been implicated in the destruction of the ozone layer.Ĭhlorine has isotopes with mass numbers ranging from 32 to 40. 
As a common disinfectant, chlorine compounds are used in swimming pools to keep them clean and sanitary. Chlorine is a powerful oxidant and is used in bleaching and disinfectants. It has a disagreeable, suffocating odor that is detectable in concentrations as low as 3.5 ppm and is poisonous. In its common elemental form (Cl 2 or "dichlorine") under standard conditions, it is a pale green gas about 2.5 times as dense as air. As the chloride ion, which is part of common salt and other compounds, it is abundant in nature and necessary to most forms of life, including humans. It is a halogen, found in the periodic table in group VII (formerly VIIa or VIIb). Template:Infobox chlorine Chlorine ( Template:IPAEng, from the Greek word 'χλωρóς' (khlôros) meaning 'green'), is the chemical element with atomic number 17 and symbol Cl.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
